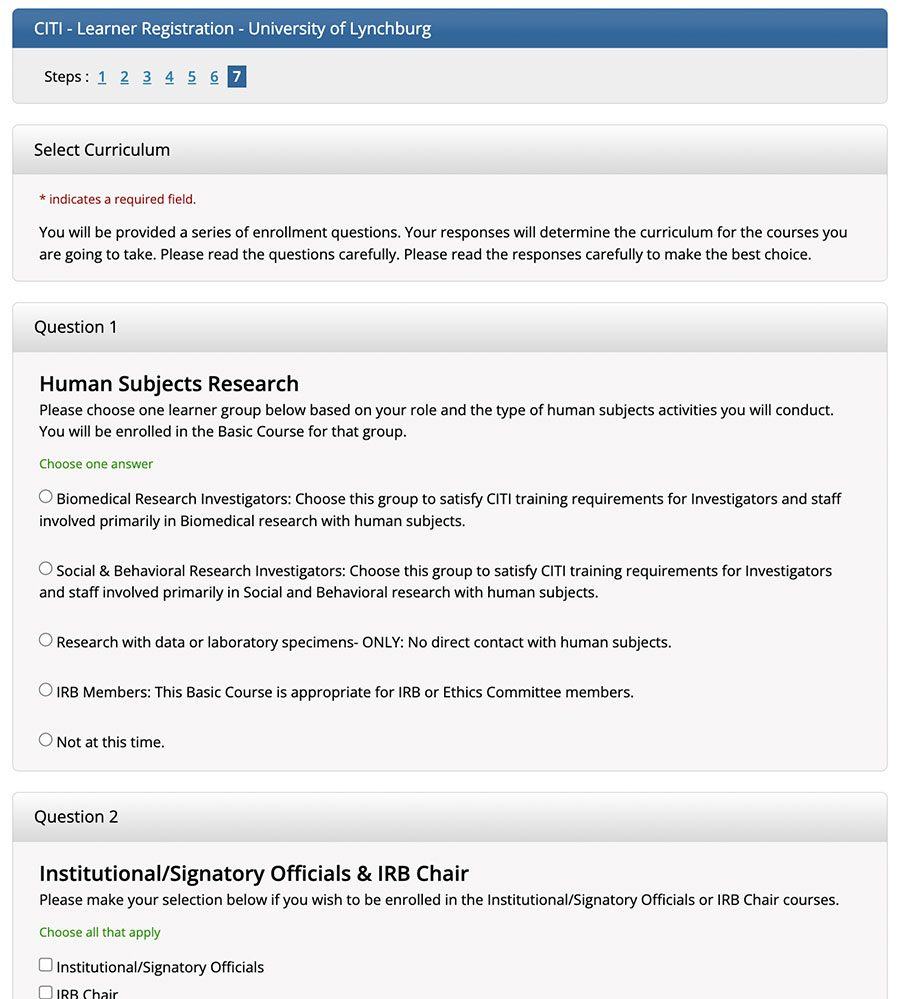
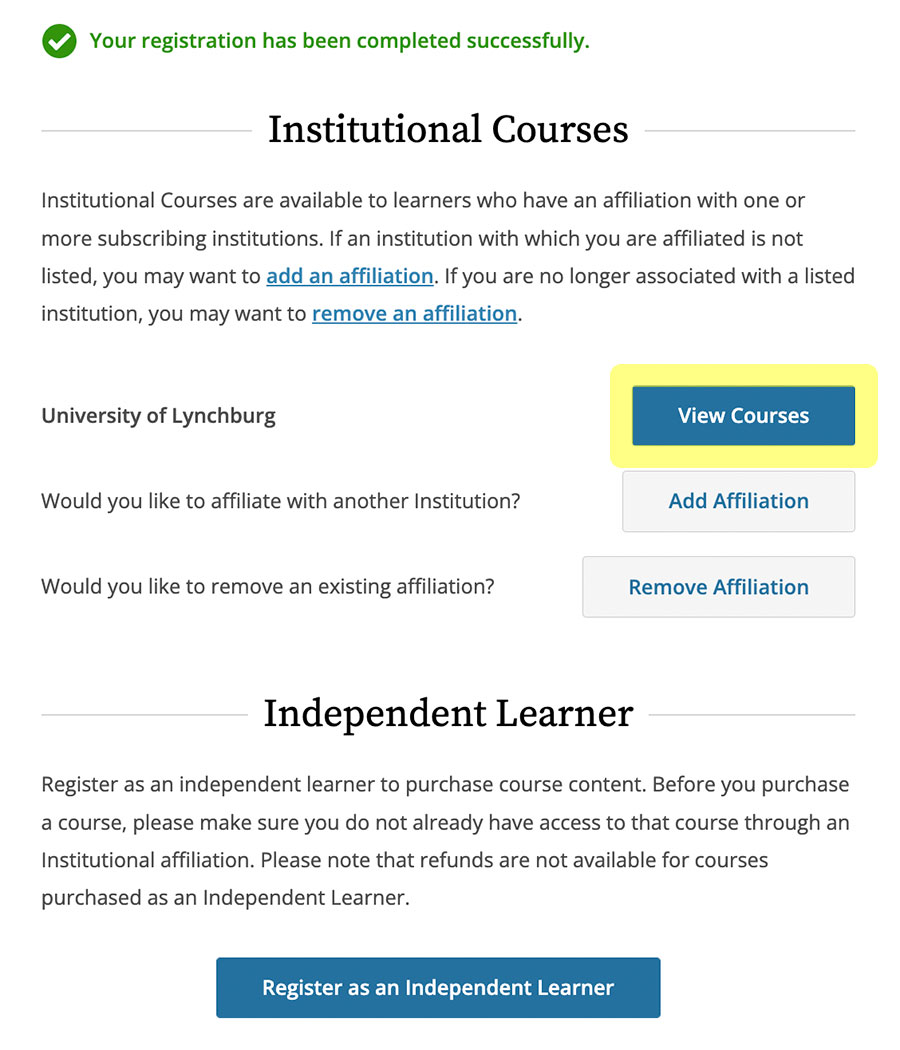
All individuals seeking IRB approval for research proposals must submit a certificate of completion for the CITI program ethics training course. In order for the certificate to be valid, the course completion date on the certificate must be within three years. A valid certificate of completion must be included in the research protocol proposal (item #11) for all research team members involved in data collection, or with access to the data. All individuals must be recertified every three years. Please note: A valid NIH certificate will be accepted through August 2021.
CITI Program Ethics Training
In order to complete the online ethics training modules, please visit the CITI Program website.
The training will take between 1 and 3 hours to complete, depending upon your reading speed, familiarity level with research ethics, and number of modules being completed.
If you are a returning user, please login with your username and password. If you have not done so already, you will need to add an affiliation to the University of Lynchburg.
If you do not have an account, you will need to register by creating a username and password (registration is free for those affiliated with the University of Lynchburg). Please see below for step-by-step instructions for CITI registration.
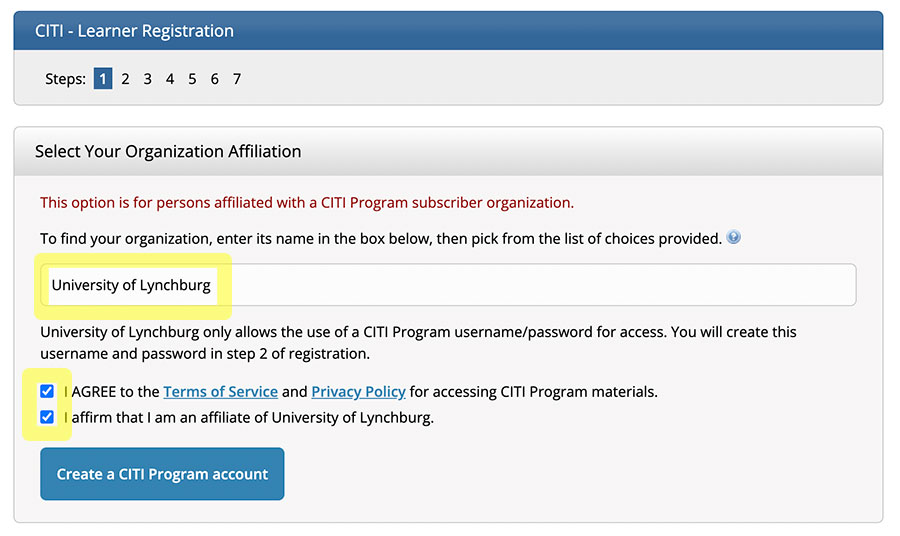
Go to www.citiprogram.org and Click “Register” in the top right hand corner.

Click the two check-boxes to agree to the terms of service and to affirm your affiliation with UL.
Next, click “Create a CITI Program account” to create username/password.
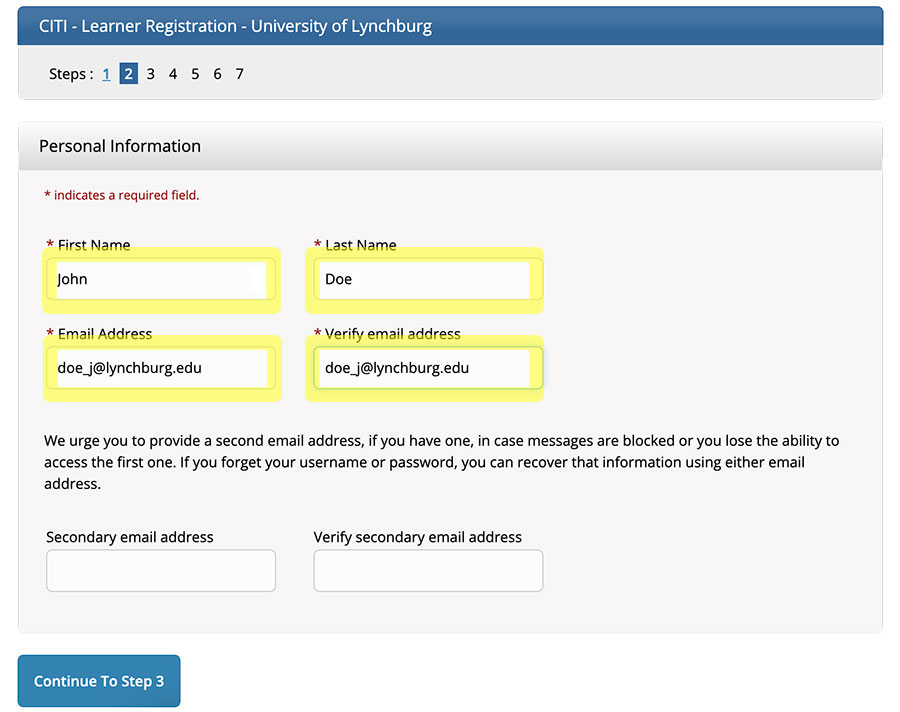
Click “Continue To Step 3.”
Click “Continue To Step 4.”
Select if you will allow additional contact after you complete your CITI coursework.
Click “Continue To Step 5.”
If not needed, select “No” and then click “Continue To Step 6.”
Click “Continue To Step 7.”
Click “Continue.”